Hello everyone, here i want to share my experience with CGM glucose measurements, food testing and diet decision-making process based on glycemic responses. I’m skipping here on topic “why glucose spikes are bad” there a lot of scientific studies confirming this.
My methodology is:
- apply CGM to measure glucose levels in high resolution with ability to export data and process it
- make isolated product tests
- produce single objective glucose response measure for each test
- build a list of foods and sort them according to responses
- exclude food which spikes even in small quantities, limit food which in a mid / high-mid range (and objectively calc limits) and do not limit food in lower mid / low.
My current setup is explained in details in my blog. In a short i’m using Freestyle Libre V1 sensor, android app Glimp to read data from sensor and export data in CSV.
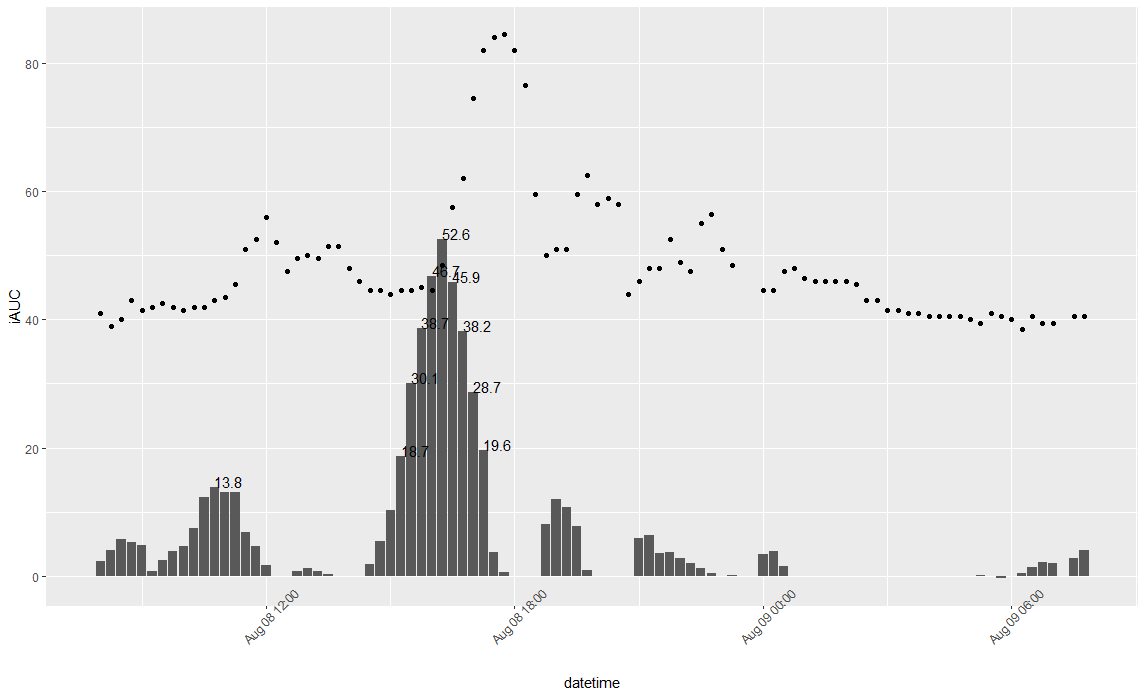
After reading some papers about glycemic index (GI) i decided that iAUC - Area Under the Curve of glucose levels 120 minutes after meal is an objective measure which represents glycemic response and may be used in predicting how much food to eat to stay in range.
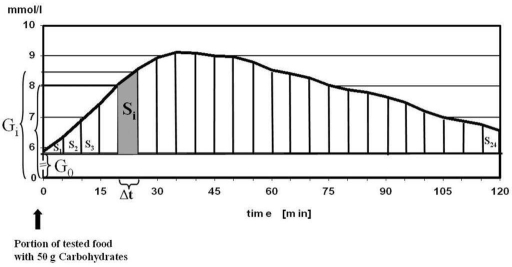
Generally speaking, iAUC is an average time-weighted glucose level change during 2 hours after meal (PPGR - postprandial glucose response) and units are same as for glucose levels.
To make different food tests comparable they should be done at same amount of carbo, for example 50g of carbo, like in this paper.
First of all, i generate ideas on which food i want to test, then find how much carbo per 100g and make a portion containing 50g of carbo. I make tests in the morning, ~1 hour after waking up and rest for 2 hours after meal, then export CGM data into CSV and calculate iAUC with R script.
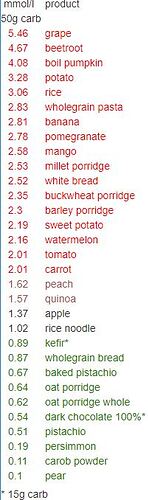
At the moment i did 30 tests and here is my results. This table is personal, different people will have different responses!
By knowing how much different products influence glucose levels i can decide how much to eat. I know that 50g of mango carbo’s (300g mango portion) increase my glucose (iAUC) by 2.58 mmol/L and by knowing that i can decide to eat half portion (150-200g) and be confident that my glucose will stay in range (generally i aim for <2-2.5mmol/L). We can see that 50g carbo from tomatoes is slightly above 2 mmol/L, but there is 5g of carbo per 100g of tomato. So eating even half kilo of tomatoes would not spike my glucose and it’s safe to eat them. Also i can see that it’s better to avoid grape and beetroot (but if there is a 1 single grape in salad, that’s fine).
Also we should take into account that
- complex mixed meals may not be a sum of separated test responses
- there is limited evidence that protein and fat may slow glucose uptake
- protein may increase glucose a bit by itself, because some glucose can be made from glucogenic amino acids
- circadian rhytms affect glucose uptake. In the morning we metabolise glucose better than before sleep
- exercise and training affect glucemic responses
- sleep quality and sickness affect glucemic responses
- glycemic resonses may change over time
- boiled carrot and fresh carrot are two different products and both needs to be tested
- multiple tests for single product are prefered, to exclude random deviations
- this approach is still rough, but better than relying just on GI from google
- there are some data that microbiome may predict glycemic responses and we can pass on hardcore testing each product one by one
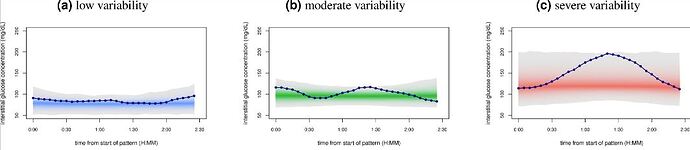
After all, how can i validate that this methooly is working for me? There is interesting paper about glucose disregulation and cluster people postprandial glucose responses as low, moderate and high variability.
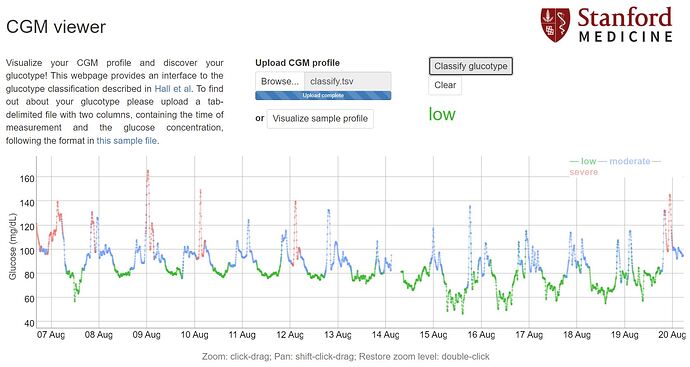
They provide a webtool for classifying glucotypes, just need few days of cgm glucose measurements formatted into tab separated values.
When i wear cgm sensor first time i got moderate group. Lowest risks were in low group (significant part of high/moderate developed T2D in long term). After some food testing and adjustings to my diet i was able to optimize my glucose responses and last 3 periods of wearing cgm i’m in low variability range. That makes me confident that i’m going into right direction.
Dont forget that this is n=1 and i may have mistakes in methodology, aims and results.
R script and raw data are available in my blog.
P.S. i dont have diabetes, just want to make health beneficial long term decisions ![]()